Page 357 - SAHCS HIVMed Journal Vol 20 No 1 2019
P. 357
Page 6 of 10 Original Research
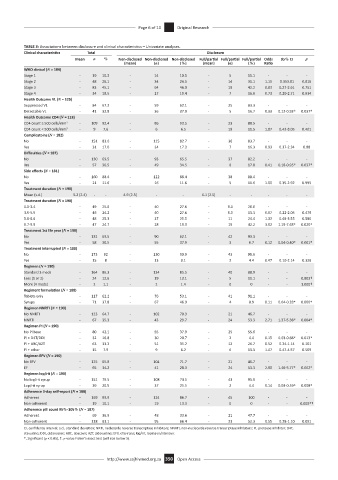
TABLE 3: Associations between disclosure and clinical characteristics – Univariate analyses.
Clinical characteristics Total Disclosure
Mean n % Non-disclosed Non-disclosed Non-disclosed Full/partial Full/partial Full/partial Odds 95% CI p
(mean) (n) (%) (mean) (n) (%) Ratio
WHO clinical (N = 184)
Stage 1 - 19 10.3 - 14 10.1 - 5 11.1 - - -
Stage 2 - 48 26.1 - 34 24.5 - 14 31.1 1.15 0.353.81 0.816
Stage 3 - 83 45.1 - 64 46.0 - 19 42.2 0.83 0.27-2.61 0.751
Stage 4 - 34 18.5 - 27 19.4 - 7 15.6 0.73 0.20-2.71 0.634
Health Outcome VL (N = 125)
Suppressed VL - 84 67.2 - 59 62.1 - 25 83.3 - - -
Detectable VL - 41 32.8 - 36 37.9 - 5 16.7 0.33 0.12-0.93* 0.037*
Health Outcome CD4 (N = 118)
CD4 count ≥ 500 cells/mm 3 - 109 92.4 - 86 93.5 - 23 88.5 - - -
CD4 count < 500 cells/mm 3 - 9 7.6 - 6 6.5 - 19 11.5 1.87 0.43-8.05 0.401
Complicatoins (N = 182)
No - 151 83.0 - 115 82.7 - 36 83.7 - - -
Yes - 31 17.0 - 24 17.3 - 7 16.3 0.93 0.37-2.34 0.88
Difficulties (N = 187)
No - 130 69.5 - 93 65.5 - 37 82.2 - - -
Yes - 57 30.5 - 49 34.5 - 8 17.8 0.41 0.18-0.95* 0.037*
Side effects (N = 181)
No - 160 88.4 - 122 88.4 - 38 88.4 - - -
Yes - 21 11.6 - 16 11.6 - 5 11.6 1.00 0.35-2.92 0.995
Treatment duration (N = 190)
Mean (s.d.) 5.2 (2.4) - - 4.9 (2.3) - - 6.1 (2.5) - - - - -
Treatment duration (N = 190)
0.0-3.4 - 49 25.8 - 40 27.6 - 9.0 20.0 - - -
3.5-5.5 - 46 24.2 - 40 27.6 - 6.0 13.3 0.67 0.22-2.05 0.479
5.6-6.6 - 48 25.3 - 37 25.5 - 11 24.4 1.32 0.49-3.55 0.580
6.7-9.9 - 47 24.7 - 28 19.3 - 19 42.2 3.02 1.19-7.63* 0.020*
Treatment 1st life year (N = 190)
No - 132 69.5 - 90 62.1 - 42 93.3 - - -
Yes - 58 30.5 - 55 37.9 - 3 6.7 0.12 0.04-0.40* 0.001*
Treatment interrupted (N = 188)
No - 173 92 - 130 90.9 - 43 95.6 - - -
Yes - 15 8 - 13 9.1 - 2 4.4 0.47 0.10-2.14 0.326
Regimen (N = 190)
Standard 3 meds - 164 86.3 - 124 85.5 - 40 88.9 - - -
Less (1 or 2) - 24 12.6 - 19 13.1 - 5 11.1 - - 0.803†
More (4 meds) - 2 1.1 - 2 1.4 - 0 0 - - 1.000†
Regiment formulation (N = 188)
Tablets only - 117 62.2 - 76 53.1 - 41 91.1 - - -
Syrups - 71 37.8 - 67 46.9 - 4 8.9 0.11 0.04-0.33* 0.000*
Regimen NNRTI (N = 190)
No NNRTI - 123 64.7 - 102 70.3 - 21 46.7 - - -
NNRTI - 67 35.3 - 43 29.7 - 24 53.3 2.71 1.37-5.38* 0.004*
Regimen PI (N = 190)
No PI base - 80 42.1 - 55 37.9 - 25 55.6 - - -
PI + D4T/DDI - 32 16.8 - 30 20.7 - 2 4.4 0.15 0.03-0.66* 0.013*
PI + ABC/AZT - 63 33.2 - 51 35.2 - 12 26.7 0.52 0.24-1.14 0.101
PI + other - 15 7.9 - 9 6.2 - 6 13.3 1.47 0.47-4.57 0.509
Regimen EFV (N = 190)
No EFV - 125 65.8 - 104 71.7 - 21 46.7 - - -
EF - 65 34.2 - 41 28.3 - 24 53.3 2.90 1.46-5.77* 0.002*
Regimen lop/rit (N = 190)
No lop/rit syrup - 151 79.5 - 108 74.5 - 43 95.6 - - -
Lop/rit syrup - 39 20.5 - 37 25.5 - 2 4.4 0.14 0.03-0.59* 0.008*
Adherence 3-day self-report (N = 188)
Adherent - 169 89.9 - 124 86.7 - 45 100 - - -
Non-adherent - 19 10.1 - 19 13.3 - 0 0 - - 0.008*†
Adherence pill count 95%-105% (N = 187)
Adherent - 69 36.9 - 48 33.6 - 21 47.7 - - -
Non-adherent - 118 63.1 - 95 66.4 - 23 52.3 0.55 0.28-1.10 0.091
CI, confidence interval; s.d., standard deviation; NRTI, nucleoside reverse transcriptase inhibitors; NNRTI, non-nucleoside reverse transcriptase inhibitors; PI, protease inhibitor; D4T,
stavudine; DDI, didanosine; ABC, abacavir; AZT, zidovudine; EFV, efavirenz; lop/rit, lopinavir/ritonavir.
*, Significant (p < 0.05), †, p-value Fisher’s exact test (cell size below 5).
http://www.sajhivmed.org.za 350 Open Access