Page 328 - SAHCS HIVMed Journal Vol 20 No 1 2019
P. 328
Page 4 of 7 Original Research
30 BMI Thinness (<-2SD) Overweight (>+1SD)
Male Female
28
26
+1SD
+1SD
24
BMI (kg/m 2 ) 22
20
18
-2SD
-2SD
16
14
8 10 12 14 16 18 20 8 10 12 14 16 18 20
Age (years)
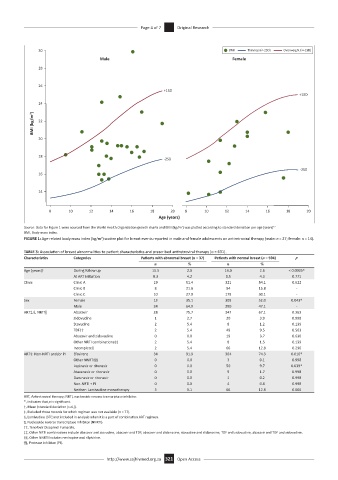
Source: Data for Figure 1 were sourced from the World Health Organization growth charts and BMI (kg/m ) was plotted according to standard deviation per age (years) 12
2
BMI, Body mass index.
FIGURE 1: Age-related body mass index (kg/m ) scatter plot for breast events reported in male and female adolescents on antiretroviral therapy (male: n = 27; female: n = 14).
2
TABLE 3: Association of breast abnormalities to patient characteristics and prescribed antiretroviral therapy (n = 631).
Characteristics Categories Patients with abnormal breast (n = 37) Patients with normal breast (n = 594) p
n % n %
Age (years)† During follow-up 15.5 2.0 14.0 2.6 < 0.0005*
At ART initiation 8.3 4.2 8.5 4.3 0.771
Clinic Clinic A 19 51.4 321 54.1 0.622
Clinic B 8 21.6 94 15.8 -
Clinic C 10 27.0 179 30.1 -
Sex Female 13 35.1 309 52.0 0.043*
Male 24 64.9 280 47.1 -
ART‡,§, NRTI¶ Abacavir 28 75.7 347 67.1 0.363
Zidovudine 1 2.7 20 3.9 0.998
Stavudine 2 5.4 8 1.2 0.139
TDF†† 2 5.4 49 9.5 0.563
Abacavir and zidovudine 0 0.0 19 3.7 0.630
Other NRTI combinations‡‡ 2 5.4 8 1.5 0.139
Incomplete§ 2 5.4 66 12.8 0.296
ART‡: Non-NRTI and/or PI Efavirenz 34 91.9 384 74.3 0.016*
Other NNRTI§§ 0 0.0 3 0.1 0.998
Lopinavir or ritonavir 0 0.0 50 9.7 0.039*
Atazanavir or ritonavir 0 0.0 9 1.7 0.998
Darunavir or ritonavir 0 0.0 1 0.2 0.998
Non-NRTI + PI 0 0.0 4 0.8 0.998
Neither: Lamivudine monotherapy 3 8.1 66 12.8 0.606
ART, Antiretroviral therapy; NRTI, nucleoside reverse transcriptase inhibitor.
*, indicates that p is significant.
†, Mean (standard deviation [s.d.]).
‡, Excluded those records for which regimen was not available (n = 77).
§, Lamivudine (3TC) not included in analysis when it is a part of combination ART regimen.
¶, Nucleoside reverse transcriptase inhibitor (NNRTI).
††, Tenofovir Disoproxil Fumarate.
‡‡, Other NRTI combinations include abacavir and stavudine, abacavir and TDF, abacavir and didanosine, stavudine and didanosine, TDF and zidovudine, abacavir and TDF and zidovudine.
§§, Other NNRTI includes nevirapine and rilpivirine.
¶¶, Protease inhibitor (PI).
http://www.sajhivmed.org.za 321 Open Access